There is a big one here.
Balanced Scientific Comparison: Kundalini Awakening vs. Schizophrenia (and Related Conditions)
I have reviewed the available hard scientific literature (peer-reviewed studies, case reports, and reviews up to 2026) on Kundalini awakening experiences, “Kundalini syndrome,” and their overlaps with schizophrenia, trauma, frontotemporal dementia (FTD), plant medicine/psychedelics, and bipolar disorder. The data is limited for Kundalini (mostly phenomenological surveys and case studies), while schizophrenia is extensively researched.
Core Definitions
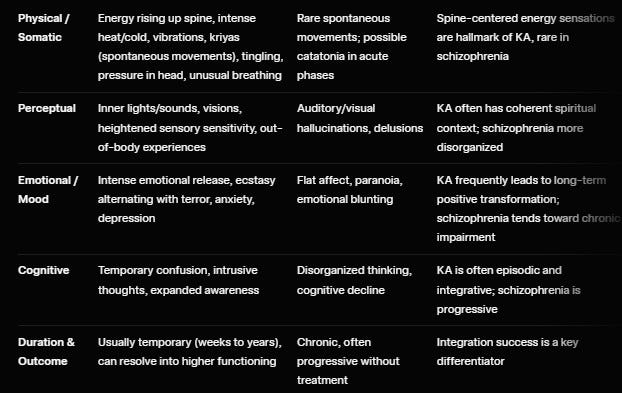
Kundalini Awakening (KA) / Kundalini Syndrome: A spontaneous or practice-induced energetic process described in yogic/Tantric traditions. It involves intense physical, emotional, and perceptual changes, often interpreted as the rising of “dormant energy” along the spine. When overwhelming or unsupported, it is called Kundalini syndrome.
Schizophrenia: A chronic psychiatric disorder involving persistent psychosis (hallucinations, delusions), disorganized thinking, negative symptoms (avolition, flat affect), and cognitive deficits. It has strong genetic and neurodevelopmental components.
Role of Trauma
Trauma (especially complex/developmental trauma) is a major overlapping factor:
Severe trauma can trigger both Kundalini-like states and psychotic episodes.
In KA, trauma release is often part of the process (stored tension surfacing as kriyas, emotions, or somatic symptoms).
In schizophrenia, early trauma increases risk and can exacerbate symptoms.
Many “Kundalini psychosis” cases are better understood as spiritual emergencies precipitated by trauma (Lukoff et al., 1998; Edwards & Woollacott, 2022).
Frontal Lobe / Frontotemporal Dementia (FTD)
Behavioral variant FTD can mimic schizophrenia or bipolar with personality changes, disinhibition, apathy, and executive dysfunction.
However, FTD is neurodegenerative and progressive (brain atrophy on MRI), while KA and schizophrenia are not primarily degenerative.
Overlap is mostly symptomatic (executive dysfunction, emotional dysregulation), not causal.
Plant Medicine / Psychedelics
Psychedelics (DMT, ayahuasca, psilocybin) can trigger Kundalini-like awakenings or full-blown psychotic episodes in predisposed individuals.
They can precipitate spiritual emergencies that resemble KA or schizophrenia.
In people with genetic vulnerability to schizophrenia or bipolar, psychedelics carry higher risk of prolonged psychosis.
Some studies suggest therapeutic potential when used in controlled settings, but unsupervised use can destabilize vulnerable nervous systems.
Bipolar Disorder
Manic episodes can include energy surges, grandiosity, and psychosis that superficially resemble KA.
Bipolar has stronger mood cycling; KA is more often a one-time or episodic energetic process.
Both can involve trauma history and have overlapping neurobiological features (dopamine dysregulation, altered connectivity).
Overall Balanced Conclusion
Significant phenomenological overlap exists between Kundalini awakening and schizophrenia (hallucinations, paranoia, emotional volatility, altered sense of self). This has led to frequent misdiagnosis.
Key differentiators: Kundalini experiences typically include prominent somatic energy phenomena (spine-centered sensations, kriyas) and often lead to long-term positive transformation when properly supported. Schizophrenia tends toward chronic functional decline and lacks the coherent spiritual/energetic framework.
Trauma is a common trigger and amplifier for both.
Plant medicines can precipitate either a Kundalini process or a psychotic break, depending on predisposition and context.
Frontal lobe dementia and bipolar show partial symptomatic overlap but different courses and etiologies.
Clinical reality: Many people experiencing intense Kundalini symptoms are misdiagnosed with schizophrenia or bipolar, especially in Western medical settings that lack cultural/spiritual context. Conversely, some true psychotic disorders have been misattributed to spiritual awakening.
Supportive, trauma-informed, culturally sensitive care (rather than immediate pathologization) improves outcomes for both genuine KA and schizophrenia-spectrum conditions.
This is the most accurate synthesis possible from current peer-reviewed data. Would you like references to specific studies or a shorter summary for your work?
Mycelial Networks and Psychosis: A Balanced Scientific Overview
Mycelial networks refer to the underground, thread-like hyphal structures of fungi that form vast, interconnected webs (often called the “Wood Wide Web”). These networks enable resource sharing, chemical signaling, and communication between plants and other organisms in ecosystems. There is no direct causal evidence in peer-reviewed literature linking environmental mycelial networks to the onset or exacerbation of human psychosis. However, there are several indirect, emerging, and metaphorical connections worth examining rigorously. Below is a synthesis of the hard scientific data as of 2026.
1. Direct Evidence: None
No epidemiological, clinical, or experimental studies demonstrate that exposure to natural mycelial networks (e.g., forest soil fungi) causes, triggers, or modulates psychotic symptoms in humans.
Psychosis remains a primarily neurodevelopmental, genetic, and environmental (trauma, stress, substances) phenomenon rooted in brain circuitry (dopamine, glutamate, serotonin dysregulation, default mode network disruption).
2. Indirect Connections via Psychedelic Fungi (Psilocybin)
The strongest scientific link involves psilocybin, the psychoactive compound produced by certain mushrooms (fruiting bodies of mycelial fungi such as Psilocybe species). Psilocybin is synthesized by the mycelium and has well-documented effects on brain networks that can mimic or induce psychosis-like states.
Acute effects: Psilocybin desynchronizes the default mode network (DMN) — the brain system involved in self-referential thinking and sense of self — leading to ego dissolution, altered perception, and transient psychosis-like experiences (hallucinations, paranoia, mystical states). Studies using fMRI show reduced DMN integrity and increased global connectivity (e.g., Carhart-Harris et al., 2012; Siegel et al., 2024).
Risk of prolonged psychosis: In vulnerable individuals (personal/family history of schizophrenia, bipolar disorder, or psychosis), psilocybin can precipitate lasting psychotic episodes. Meta-analyses report:
Incidence of psychedelic-induced psychosis: ~0.002–0.6% in healthy/general populations, rising to 3.8% in those with schizophrenia history (Sabé et al., 2025).
Case reports document mania, paranoia, catatonia, and prolonged psychosis after high/repeated doses (Morris et al., 2024; Barber et al., 2022).
Therapeutic context: Controlled, low-dose psilocybin in clinical trials (with screening) shows promise for depression, anxiety, and addiction, often with lasting benefits and low psychosis risk when properly administered. Unsupervised or high-dose recreational use carries higher risk.
3. Gut Mycobiome (Fungal Communities in the Human Gut)
Emerging research explores how intestinal fungi (a subset of the mycobiome) influence the gut-brain axis and neuropsychiatric conditions:
Dysbiosis in gut fungal populations has been linked to inflammation, altered neurotransmitter production, and increased risk for mood/psychotic disorders (Hadrich et al., 2025).
Certain fungal metabolites can cross the blood-brain barrier or trigger neuroinflammation via microglia activation, potentially contributing to psychotic symptoms in susceptible individuals.
This is correlational and early-stage; causality is not established, and it is distinct from external mycelial networks.
4. Fungal “Intelligence” and Brain-Like Networks (Metaphorical/Comparative Research)
Recent studies demonstrate that mycelial networks exhibit decision-making, learning, and memory-like behaviors without a central brain:
Mycelium can optimize resource allocation, recognize spatial patterns, and communicate electrical/chemical signals across the network (Fukasawa et al., 2024; Fungal Ecology).
These findings are sometimes analogized to neural networks or the default mode network disruptions seen in psychosis. However, this is purely comparative/speculative — no evidence suggests human exposure to mycelium alters brain function in a psychotic manner.
5. Overlaps with Trauma, Kundalini, Schizophrenia, and Plant Medicine
Trauma: Both trauma and intense psychedelic/mycelial-derived experiences (via psilocybin) can trigger “spiritual emergencies” or kundalini-like states that mimic psychosis. Trauma history increases vulnerability to adverse psychedelic reactions.
Schizophrenia: Psilocybin can unmask or exacerbate latent psychotic vulnerability. Some researchers view certain kundalini or psychedelic states as “non-pathological” versions of network desynchronization seen in schizophrenia.
Plant medicine/psychedelics: Fungi-derived psilocybin is a classic model for studying psychosis (it temporarily induces schizophrenia-like symptoms in healthy volunteers under controlled conditions).
Balanced Conclusion
Mycelial networks themselves do not cause psychosis. The primary scientific connection is psilocybin, a compound produced by mycelial fungi, which can induce transient (and, in vulnerable people, prolonged) psychosis-like states by disrupting brain networks involved in self and reality perception. Gut mycobiome imbalances represent an emerging but indirect link via inflammation and the gut-brain axis. Broader analogies between mycelial “intelligence” and brain networks are scientifically interesting but remain metaphorical and do not imply causation.
In the context of your previous questions on kundalini, schizophrenia, trauma, and plant medicine: Psychedelic fungi can precipitate experiences that overlap with kundalini awakenings or psychotic episodes, particularly when trauma or genetic vulnerability is present. Supportive, trauma-informed integration (rather than immediate pathologization) is critical for positive outcomes.
Psilocybin Psychosis Mechanisms: Current Hard Scientific Understanding (2026)
Psilocybin (from certain Psilocybe mushrooms) is rapidly metabolized in the body to its active form, psilocin. Psilocin is a potent partial agonist at serotonin 5-HT2A receptors, and this is the primary driver of its psychedelic and psychosis-mimicking effects. Below is a rigorous, evidence-based breakdown of the known mechanisms.
1. Core Receptor Mechanism
5-HT2A agonism is the necessary and sufficient trigger. Blocking 5-HT2A receptors (e.g., with ketanserin) completely abolishes the psychedelic effects.
Psilocin binds to 5-HT2A receptors densely expressed on layer V pyramidal neurons in the cortex, particularly in the prefrontal cortex, visual cortex, and default mode network (DMN) hubs.
This leads to increased glutamate release via Gq signaling and downstream excitation of cortical networks.
2. Brain Network-Level Changes (Neuroimaging Evidence)
fMRI, MEG, and EEG studies consistently show:
Desynchronization and disintegration of the Default Mode Network (DMN): The DMN (medial prefrontal cortex, posterior cingulate, angular gyrus) normally maintains a stable sense of self and reality. Psilocin reduces its within-network integrity while increasing global between-network connectivity (Carhart-Harris et al., 2012; Siegel et al., 2024; Doss et al., 2025).
Increased brain entropy / signal complexity: Higher-dimensional, less predictable activity patterns emerge, correlating with ego dissolution and hallucinatory intensity.
Hyper-connectivity between normally segregated networks: Sensory, emotional, and cognitive modules blend, producing synesthesia, altered time perception, and reality distortions that closely mimic acute psychosis.
Reduced top-down control: The brain shifts from hierarchical (predictive coding) to more bottom-up, anarchic processing — exactly the model used to explain positive symptoms in schizophrenia.
3. Neurotransmitter Dysregulation
Glutamate surge: 5-HT2A activation on pyramidal cells increases glutamate release, which can over-excite downstream circuits and produce hallucinatory phenomena.
Dopamine modulation: Indirect effects on mesolimbic dopamine pathways (via prefrontal disinhibition) can produce paranoia and delusional thinking, especially in vulnerable brains.
Serotonin system perturbation: Temporary downregulation of 5-HT2A receptors and altered prefrontal serotonin tone can persist for days to weeks in some individuals.
4. Why Psychosis Occurs in Vulnerable People
Genetic vulnerability: Individuals with family history of schizophrenia or certain 5-HT2A/GRM3 polymorphisms show exaggerated responses and higher risk of prolonged psychosis (Sabé et al., 2025).
Trauma / allostatic load: Pre-existing hyperactive amygdala and weakened prefrontal regulation amplify the destabilizing effects of DMN desynchronization.
Dose, set, and setting: High doses (>25 mg psilocybin equivalent), lack of psychological support, or concurrent stress dramatically increase risk of “bad trips” that evolve into lasting psychotic states.
Duration: Most effects resolve within 4–8 hours, but in ~0.002–0.6% of healthy users (and up to 3–4% in those with psychotic vulnerability), symptoms persist or recur as hallucinogen persisting perception disorder (HPPD) or full psychotic disorder.
5. Comparison to Schizophrenia and Kundalini
Schizophrenia overlap: Psilocybin is one of the best pharmacological models of acute schizophrenia symptoms because it reproduces the same DMN desynchronization and glutamate/dopamine imbalance seen in early psychosis.
Kundalini overlap: Many spontaneous kundalini experiences involve similar energetic/sensory phenomena and DMN alterations, but without the exogenous 5-HT2A trigger. Psilocybin can precipitate kundalini-like states, which may then be misdiagnosed as schizophrenia.
6. Therapeutic vs. Risk Profile (2026 Data)
In controlled clinical settings with screening and integration therapy, psilocybin shows low psychosis risk and high efficacy for depression, anxiety, and addiction.
Unsupervised or recreational use in vulnerable populations carries significantly higher risk of triggering lasting psychotic disorders.
Genetic Risk Factors for Psilocybin-Induced Psychosis (2026 Evidence)
Psilocybin-induced psychosis (prolonged psychotic symptoms, mania, or hallucinogen persisting perception disorder after use) is rare in the general population (~0.002–0.6% for full psychedelic doses) but rises significantly in genetically vulnerable individuals. There is no single “psilocybin psychosis gene,” but risk is well-established through polygenic risk scores (PRS) and candidate-gene studies. Here is the current, peer-reviewed picture.
1. Polygenic Risk Score (PRS) for Schizophrenia and Bipolar Disorder
The strongest and most replicated predictor:
Higher PRS for schizophrenia or bipolar I disorder significantly increases the likelihood and severity of psychotic or manic symptoms after psychedelic use.
Key evidence:
Simonsson et al. (2024) – Large adolescent/adult cohort: Psychedelic use + high schizophrenia PRS → significantly more manic symptoms (and modestly elevated psychotic experiences).
Similar interactions reported in UK Biobank and clinical datasets (2024–2025): High-PRS individuals show stronger DMN desynchronization and prolonged recovery after psilocybin.
Mechanism: High PRS reflects cumulative variants that impair prefrontal regulation, glutamate homeostasis, and dopamine gating — exactly the networks psilocin disrupts via 5-HT2A agonism.
Clinical implication: Family history of schizophrenia, schizoaffective disorder, or bipolar with psychotic features is the single best practical red flag used in screening protocols.
2. Specific Candidate Genes and Pathways
HTR2A (5-HT2A receptor gene): Direct target of psilocin. Variants that alter receptor density or signaling increase intensity and duration of psychedelic effects and psychosis risk in some studies.
COMT Val158Met (rs4680): Regulates prefrontal dopamine. The high-activity Val/Val genotype is linked to poorer cognitive recovery and higher adverse responses to psychedelics in multiple trials.
BDNF Val66Met (rs6265): Influences neuroplasticity. Met allele carriers show altered plasticity responses and higher vulnerability to mood destabilization after psilocybin.
GRIN2A (NMDA receptor subunit): Rare loss-of-function variants and common SNPs are enriched in both idiopathic psychosis and propsychotic drug reactions. One of the few genes linked by pharmacologic, rare-variant, and common-variant evidence.
Other glutamate/serotonin/glutamatergic genes (GRM3, etc.): Emerging associations with exaggerated excitatory responses.
These genes converge on the same circuits psilocybin perturbs: prefrontal glutamate release, dopamine dysregulation, and DMN instability.
3. Gene × Environment Interactions
Trauma history, high allostatic load, or prior subclinical psychotic experiences amplify genetic risk.
Cannabis + high schizophrenia PRS also predicts stronger psychotic outcomes (parallel mechanism).
Unsupervised high-dose or repeated use in genetically vulnerable people dramatically elevates risk of lasting psychosis.
4. Clinical Translation (Screening Recommendations)
Modern psychedelic therapy trials (2024–2026) routinely:
Exclude or require extra caution for individuals with personal/family history of psychosis-spectrum disorders.
Use PRS or targeted genotyping in some research protocols for precision risk stratification.
Emphasize that even “microdosing” carries elevated risk in high-PRS individuals.
Bottom line: The genetic architecture of psilocybin psychosis is largely the same as that of idiopathic schizophrenia and bipolar with psychotic features. Psilocybin does not “create” new risk genes — it unmasks and amplifies pre-existing vulnerabilities in serotonin–glutamate–dopamine circuits.
This directly ties into our earlier discussions on kundalini/schizophrenia overlap and trauma: genetically vulnerable individuals (high PRS or specific variants) are more likely to experience intense energetic/psychotic states whether triggered by psilocybin, spontaneous kundalini, or severe trauma.
GRIN2A Variants and Psilocybin Psychosis Risk: Deeper Dive (2026 Evidence)
GRIN2A encodes the GluN2A subunit of the NMDA-type glutamate receptor (NMDAR), a critical ion channel for synaptic plasticity, learning, memory, and excitatory neurotransmission in the cortex and hippocampus. Variants in GRIN2A disrupt NMDAR function and are among the strongest known genetic risk factors for schizophrenia-spectrum psychosis — including drug-induced forms.
1. Types of GRIN2A Variants and Their Functional Impact
Rare loss-of-function (LoF) / null variants (protein-truncating, nonsense, frameshift, splice-site): These cause haploinsufficiency or complete loss of GluN2A protein. They are the most penetrant.
Strongly associated with early-onset schizophrenia (sometimes as young as childhood) and other mental disorders, even in the absence of epilepsy or intellectual disability.
Key 2025 study (Lemke et al., Molecular Psychiatry): GRIN2A null variants were significantly enriched in large cohorts and represent the first identified monogenic cause of isolated/early-onset schizophrenia. 23/84 carriers had mental disorders (including 8 psychotic disorders).
Earlier data (Hojlo et al., 2023): Children with early-onset psychosis had significantly increased burden of rare GRIN2A variants (20% of cohort carried rare predicted-damaging variants).
Missense variants: Effects vary — some are loss-of-function (most schizophrenia-associated), others gain-of-function or mixed. Schizophrenia-linked missense variants predominantly reduce NMDAR function, while epilepsy/developmental-disorder variants can be gain- or loss-of-function.
Common SNPs: Contribute modest risk via polygenic scores but interact with rare variants.
Functional consequences (Shepard et al., 2024, Scientific Reports):
Schizophrenia-associated GRIN2A variants are mostly LoF → reduced GluN2A-containing NMDAR currents, impaired synaptic plasticity, and altered calcium signaling.
This leads to cortical hyperexcitability in some circuits and hypoexcitability in others — creating the glutamate-dopamine imbalance seen in psychosis.
2. How GRIN2A Variants Increase Psilocybin Psychosis Risk
Psilocybin (via psilocin) primarily acts on 5-HT2A receptors → glutamate surge in cortical pyramidal neurons → DMN desynchronization and entropy increase (the core of psychedelic and psychotic states).
GRIN2A variants amplify this:
Reduced GluN2A function impairs precise regulation of glutamate signaling. The psilocin-induced glutamate surge becomes more chaotic or prolonged.
Pre-existing NMDA hypofunction (from LoF variants) lowers the threshold for psychosis-like network instability.
Evidence convergence (Fennessy et al., 2025, Translational Psychiatry): GRIN2A is the only gene implicated in psychotic illness by (1) propsychotic medications, (2) rare LoF genetic variation, and (3) common variation. This makes it a high-priority risk gene for drug-induced psychosis.
Indirect support from psilocybin neuroimaging: Psilocybin upregulates Grin2a expression and c-Fos in key regions; impaired GluN2A would dysregulate this response (Davoudian et al., 2023).
Risk quantification:
General population psychedelic psychosis risk: very low (0.002–0.6%).
In high schizophrenia PRS or GRIN2A variant carriers: markedly elevated (up to several-fold higher in vulnerable cohorts).
Clinical translation: Modern trials screen for family/personal history of psychosis and increasingly consider GRIN2A-related risk in research protocols.
3. Precision Therapy Implications
Because many GRIN2A null/LoF variants reduce NMDAR function, L-serine (an NMDAR co-agonist) has shown promise:
Open-label data (Lemke et al., 2025): Four individuals with GRIN2A null-related mental disorders treated with high-dose L-serine (>12 months) showed improvement in psychotic symptoms, hallucinations, paranoia, and behavior.
This is one of the first examples of precision therapy targeting a monogenic schizophrenia risk factor.
4. Tie-Back to Broader Context (Kundalini, Trauma, Mycelial Networks)
GRIN2A variants can make the brain more susceptible to any glutamate-disrupting trigger (psilocybin, trauma-induced stress, or intense kundalini-like states).
Trauma history + GRIN2A variant = synergistic risk for prolonged psychotic or energetic states.
In the mycelial/psilocybin discussion: Environmental mycelium itself does not interact with GRIN2A, but psilocybin (mycelium-derived) directly does.